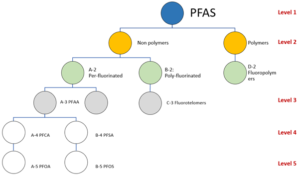
Per- and Poly-Fluoro Alkyl Substances (PFAS) are a universe of several thousand manufactured fluorinated organic chemicals with very varying chemical and physical properties (Figure 1). Some are still in use, some are present in legacy fire fighting systems and some have been banned.
This short article was initially intended as an overview of the land contamination risk management aspects of PFAS but ended up using all the words AGS Magazine articles run to in exploring definitions and naming conventions for PFAS.
Figure 1 PFAS hierarchy (only terms used in this article are shown) (After colour conventions of ITRC[1] and OECD Level numbering)
Definitions
Definitions matter[2] especially when regulations apply to named substances. The OECD and the Environment Agency[3] consider almost any chemical with at least one fully (per-) fluorinated methyl (–CF3) or methylene (–CF2–) group as a PFAS (OECD, 2021)[4]. Under this definition, the simplest PFAS is tetrafluoromethane (CF4), also known as carbon tetrafluoride or R-14 (a low temperature refrigerant).
Figure 2 Fully fluorinated methyl and methylene groups. Black = carbon; green = fluorine; grey = covalent bond with the rest of the (unshown) alkyl chain
The US EPA’s Office of Pollution Prevention and Toxics definition is narrower: PFAS have at least two adjacent carbon atoms, where one carbon is fully and the other at least partially fluorinated. This would exclude CF4 and about 40% of the substances that would meet the OECD definition.
The US Congress[5] definition sat in between the OECD and US EPA definitions: “perfluoroalkyl and polyfluoroalkyl substances that are man-made chemicals with at least one fully fluorinated carbon atom.”
OECD recognised that organisations may develop their own working definition of PFAS to meet specific needs. OECD even provided an approach based on molecular structural traits to organisations to make their own categorization in a coherent and consistent manner. The OECD highly recommends the context and rationale for such definitions be transparent to avoid confusion.
Table 1 PFAS related acronyms used in this article
| Acronym | Meaning |
| PFAS | Per- and PolyFluoroalkyl Substances |
| AFFF | aqueous film forming foams |
| PFOA | Perfluoro octanoic acid |
| PFOS | Perfluorooctane sulfonic acid |
| PFNA | Perfluorononanoic acid |
| PFCA | Perfluoro carboxylates |
| PFSA | Perfluoroalkane sulfonates |
| PFAA | Perfluoroalkyl acid |
| PFHxS | Perfluorohexanesulfonic acid |
| PASF | Perfluoroalkane sulfonyl fluoride |
What’s in a name?
There are three parts to the name of individual PFAS: the first tells you whether it is Poly- or Per-Fluorinated, the second tells you how many carbons in the alkyl chain (Hx = 6; Hp = 7; O = 8; N = 9) and the third what the functional group at the end of the molecule away from the fluorinated chain is (sulfonic acid = SA; octanoic acid = OA).
Long and short chain PFAS have significantly different properties – affecting the risks they pose and their amenity to different remediation strategies. According to the OECD (2013), “long-chain PFAS” are:
(i) Perfluoro carboxylates (PFCAs) with 7 and more fully fluorinated carbons, such as PFOA (8 carbons) and PFNA (9 carbons);
(ii) perfluoroalkyl sulfonic acids (PFSAs) with 6 and more fully fluorinated carbons, such as PFHxS (6 carbons) and PFOS (8 carbons); and
(iii) precursors such as PASF- and fluorotelomer-based compounds that can degrade to long-chain PFCAs or PFSAs.
Human health, ecological and controlled waters risk assessments reflect the fate, transport and for the first two the toxicity of the hazard.
The most studied PFAAs to date are PFOA and PFOS – the primary PFAS focus of many site investigations. However they are not the only PFAS and they cannot be used as surrogates for all PFAS.
Long and short chain PFAS behave in different ways. Short chain PFAS are very mobile, soluble and have low adsorption potential. It has also been recognised that PFAS have broad toxicity[6].
The C8 Science Panel[7], that featured in the legal case depicted in the Dark Waters film, determined that a “probable link” exists between C8 (PFOA) and the following 6 diseases:
- Kidney Cancer.
- Testicular Cancer.
- Ulcerative Colitis.
- Thyroid Disease.
- Pregnancy Induced Hypertension (including preeclampsia)
- Hypercholesterolemia
Discussion
We have been here before. About 30 years ago the dawning realisation that petroleum hydrocarbons were too broad a universe to have their varied fate, transport and toxicity represented by a single “total petroleum hydrocarbon” analysis led to the “TPHCWG” working group that gave us the Equivalent Carbon hydrocarbon fraction method of assessing the risks to human health from petroleum hydrocarbons.
The current body of scientific evidence clearly indicates that there are real, present, and significant hazards associated with specific PFAS – not least PFOS and PFOA – but significant gaps remain related to the impacts of other PFAS on human health and in the environment[8].
The time has now come for us to realise that PFAS may be a universe of substances but they are too diverse to be considered together or to consider individual substances – such as PFOS or PFOA – as surrogates in the way that we have been able to use benzo(a)pyrene as a surrogate for polyaromatic hydrocarbons (PAH).
Understanding and dealing with the societal challenge that is PFAS will involve a multi-disciplinary approach: chemists to resolve behaviour and develop analytical methods, toxicologists to establish dose-response relationships, geologists to predict subsurface fate and transport, engineers to implement remediation.
[1] https://pfas-1.itrcweb.org/2-pfas-chemistry-and-naming-conventions-history-and-use-of-pfas-and-sources-of-pfas-releases-to-the-environment-overview/
[2] https://www.sciencepolicyjournal.org/uploads/5/4/3/4/5434385/dean_adejumo_caiati_etal_jspg_v16.pdf
[3] https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1012230/Poly-_and_perfluoroalkyl_substances_-sources_pathways_and_environmental_data_-_report.pdf
[4] https://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?cote=ENV/CBC/MONO(2021)25&docLanguage=En
[5] https://www.congress.gov/116/bills/s1790/BILLS-116s1790enr.pdf
[6] https://www.sciencepolicyjournal.org/uploads/5/4/3/4/5434385/dean_adejumo_caiati_etal_jspg_v16.pdf
[7] http://www.c8sciencepanel.org/
[8] https://www.epa.gov/system/files/documents/2021-10/pfas-roadmap_final-508.pdf
Article provided by Paul Nathanail, Director, LQM